Mucosal surfaces, including those found at the intestines, mouth, nasal passages, and airways, are directly exposed to the external environment and are particularly susceptible to pathogenic bacteria. Human mucosal surfaces maintain numerous defense mechanisms, both by secreting antimicrobial and immune-stimulating molecules, as well as by maintaining a beneficial commensal microbial community that aids in the prevention of infection [1]. Interestingly, Barr et al recently reported a model (the bacteriophage adherence to mucus [BAM] model) for how phages may also play an important role in mucosal immunity [2]. This is important because it outlines a medically relevant, previously unrecognized tripartite symbiosis between humans (and other metazoans), bacteria, and phages, whereby phages will control bacterial populations to preserve human mucosal health, which is in turn beneficial for both commensal bacteria and phages. This report also provides us with a new understanding of how phages may interact with their environment, how phages can affect human immunity, and how we might use phages for therapeutic interventions at mucosal surfaces.
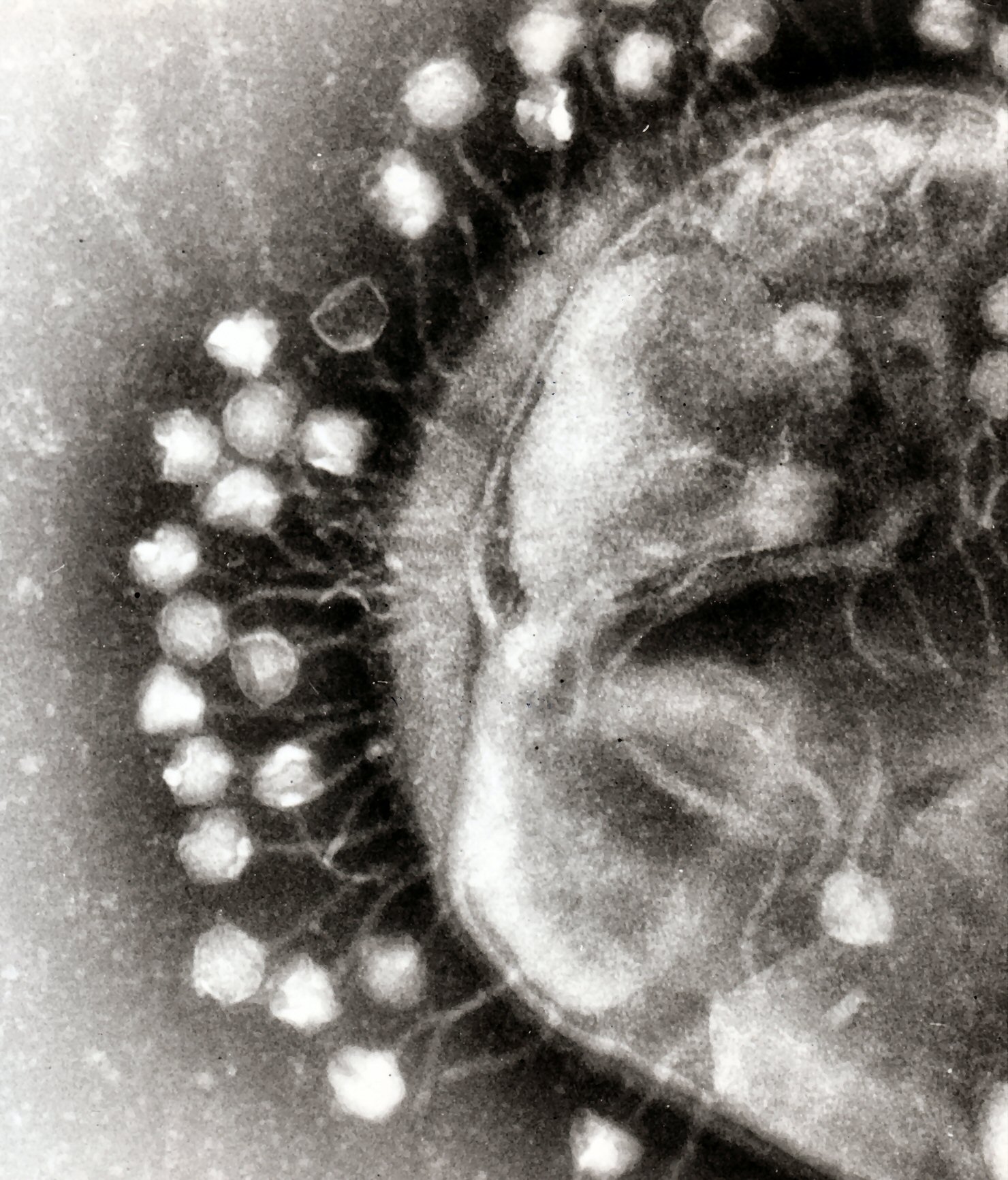
 The BAM model proposed by Barr et al (see figure to left) suggests that phages persist longer in mucous environments, compared to other environments, because they have Ig-like domains (immunoglobulin-like domains that bind certain motifs, similar to how antibodies bind to certain motifs) which bind to mucin glycoproteins. The increased phage persistence results in higher concentrations of phages at the mucus layer, thereby providing a greater chance that phages will infect and destroy a bacterial pathogen that enters the mucus. The group supports their model by performing a variety of experiments that use T4 bacteriophage, E. coli, and mucus secreting human cells.
The BAM model proposed by Barr et al (see figure to left) suggests that phages persist longer in mucous environments, compared to other environments, because they have Ig-like domains (immunoglobulin-like domains that bind certain motifs, similar to how antibodies bind to certain motifs) which bind to mucin glycoproteins. The increased phage persistence results in higher concentrations of phages at the mucus layer, thereby providing a greater chance that phages will infect and destroy a bacterial pathogen that enters the mucus. The group supports their model by performing a variety of experiments that use T4 bacteriophage, E. coli, and mucus secreting human cells.